If you're a member of the military, then you're likely familiar with the Da Form 7539. This is the form that's used to request leave from duty, and it's important that you complete it correctly if you want your leave to be approved. In this blog post, we'll provide a brief overview of the Da Form 7539 so that you know what to expect when completing it. We'll also provide some tips on how to ensure that your leave is approved. Keep in mind that this information is for informational purposes only and should not be taken as legal advice. If you have specific questions about the Da Form 7539, we suggest consulting with an attorney or legal professional. Thanks for reading!
| Question | Answer |
|---|---|
| Form Name | Da Form 7539 |
| Form Length | 2 pages |
| Fillable? | No |
| Fillable fields | 0 |
| Avg. time to fill out | 30 sec |
| Other names | SUBMITTER, OTSG, IMSL, 9V1 |
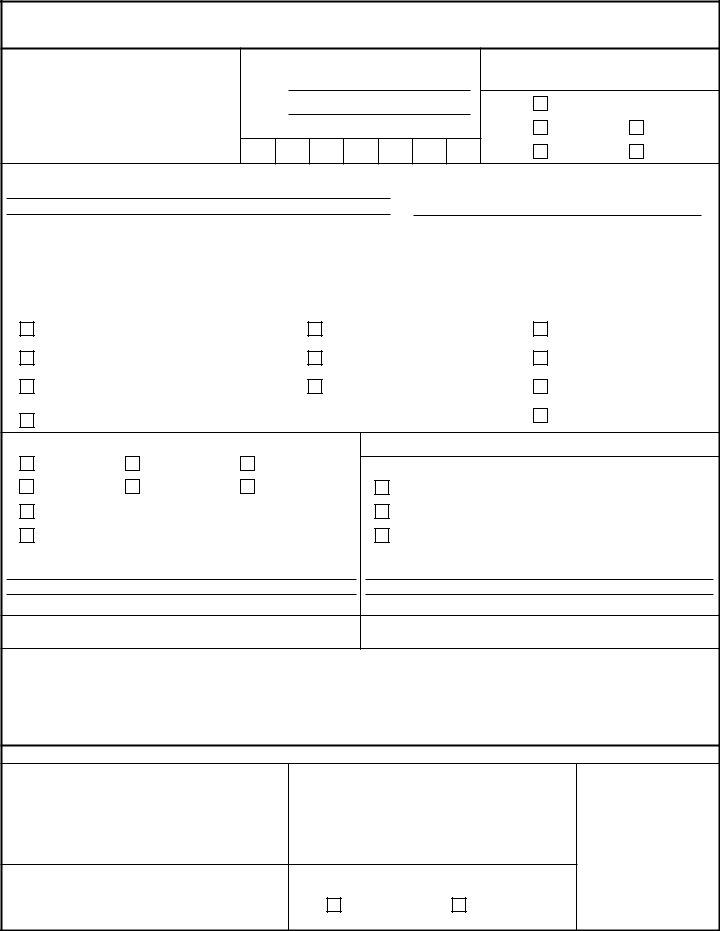
REQUEST FOR VETERINARY LABORATORY TESTING & FOOD SAMPLE RECORD
For use of this form, see AR
1 . FROM:
2 . POINT OF CONTACT:
Name:
Phone:
Station Identification Number:
-
3 . CONTROL NUMBER:
4 . TO: |
VETCOM FADL |
|
|
VLE |
BAHRAIN |
|
HAWAII |
KOREA |
5 . PRODUCER/MANUFACTURER (Name, Address and Phone):
|
|
|
|
|
ESTABLISHMENT # / PLANT CODE (IMSL, USDA, etc.) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
VC # |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
6 . REASON FOR SUBMISSION: |
|
|
|
Sanitation Audits |
|
||
|
|
|
|
|
|||
|
Suspected foodborne illness |
Destination monitoring program |
Initial |
|
|||
|
(contact laboratory prior to submission) |
|
|
|
|
|
|
|
Suspected foreign material/object |
Contract compliance |
Special |
|
|||
|
Customer return/complaint |
Proximate analysis |
Directed routine |
|
|||
|
(provide synopsis of incident/problem and local |
|
|
|
|
|
|
|
inspection results in the Remarks section below ). |
|
|
|
Routine |
|
|
|
OTHER (Specif y): |
|
|
|
|
||
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
7 . SAMPLES SELECTED FROM:
DECA |
MWR |
Exchange |
Exchange vendor |
Commercial establishment
OTHER:
PLANT
Prime vendor
8 . DATE SAMPLE(S) SELECTED: |
THRU |
9 . SHIPMENT TEMPERATURE CONDITIONS:
Room temperature
Frozen
Chilled - include 1 temperature pilot per shipping container
10 . INSPECTOR' S SIGNATURE
11 . ACCOUNTABLE OFFICER' S SIGNATURE
12 . REMARKS (use additional paper if necessary):
FOR LABORATORY USE ONLY
SHIPPING CARRIER TRACKING NUMBER: |
LABORATORY REPORT NUMBER: |
SAMPLE(S) FOR ANALYSIS BY:
RECEIPT TEMPERATURE: |
|
CHEMISTRY |
MICROBIOLOGY |
|
|
|
|
RECEIVED:
DA FORM 7539, FEB 2005
APD 9V1.000
PAGE 1 OF 2

13 . SAMPLE INFORMATION (Complete as much information as is available): |
LAB REPORT # |
|
||
|
|
|
|
|
SAMPLE NUMBER 1 |
|
FOR LABORATORY USE ONLY |
|
|
|
|
|
|
|
|
|
|
|
|
SUBMITTER SAMPLE NUMBER |
SAMPLE DESCRIPTION |
|
|
BRAND NAME |
|
|
|
|
|
UNIVERSAL PRODUCT CODE (UPC) |
PRODUCT CODE |
|
|
SAMPLE WEIGHT/VOLUME |
|
|
|
|
|
QUANTITY SUBMITTED |
UNIT OF ISSUE |
TOTAL COST |
DISPOSITION |
|
|
|
|
|
|
SAMPLE NUMBER 2 |
|
FOR LABORATORY USE ONLY |
|
|
|
|
|
|
|
|
|
|
|
|
SUBMITTER SAMPLE NUMBER |
SAMPLE DESCRIPTION |
|
|
BRAND NAME |
|
|
|
|
|
UNIVERSAL PRODUCT CODE (UPC) |
PRODUCT CODE |
|
|
SAMPLE WEIGHT/VOLUME |
|
|
|
|
|
QUANTITY SUBMITTED |
UNIT OF ISSUE |
TOTAL COST |
DISPOSITION |
|
|
|
|
|
|
SAMPLE NUMBER 3 |
|
FOR LABORATORY USE ONLY |
|
|
|
|
|
|
|
|
|
|
|
|
SUBMITTER SAMPLE NUMBER |
SAMPLE DESCRIPTION |
|
|
BRAND NAME |
|
|
|
|
|
UNIVERSAL PRODUCT CODE (UPC) |
PRODUCT CODE |
|
|
SAMPLE WEIGHT/VOLUME |
|
|
|
|
|
QUANTITY SUBMITTED |
UNIT OF ISSUE |
TOTAL COST |
DISPOSITION |
|
|
|
|
|
|
SAMPLE NUMBER 4 |
|
FOR LABORATORY USE ONLY |
|
|
|
|
|
|
|
|
|
|
|
|
SUBMITTER SAMPLE NUMBER |
SAMPLE DESCRIPTION |
|
|
BRAND NAME |
|
|
|
|
|
UNIVERSAL PRODUCT CODE (UPC) |
PRODUCT CODE |
|
|
SAMPLE WEIGHT/VOLUME |
|
|
|
|
|
QUANTITY SUBMITTED |
UNIT OF ISSUE |
TOTAL COST |
DISPOSITION |
|
|
|
|
|
|
SAMPLE NUMBER 5 |
|
FOR LABORATORY USE ONLY |
|
|
|
|
|
|
|
|
|
|
|
|
SUBMITTER SAMPLE NUMBER |
SAMPLE DESCRIPTION |
|
|
BRAND NAME |
|
|
|
|
|
UNIVERSAL PRODUCT CODE (UPC) |
PRODUCT CODE |
|
|
SAMPLE WEIGHT/VOLUME |
|
|
|
|
|
QUANTITY SUBMITTED |
UNIT OF ISSUE |
TOTAL COST |
DISPOSITION |
|
|
|
|
|
|
SAMPLE NUMBER 6 |
|
FOR LABORATORY USE ONLY |
|
|
|
|
|
|
|
|
|
|
|
|
SUBMITTER SAMPLE NUMBER |
SAMPLE DESCRIPTION |
|
|
BRAND NAME |
|
|
|
|
|
UNIVERSAL PRODUCT CODE (UPC) |
PRODUCT CODE |
|
|
SAMPLE WEIGHT/VOLUME |
|
|
|
|
|
QUANTITY SUBMITTED |
UNIT OF ISSUE |
TOTAL COST |
DISPOSITION |
|
|
|
|
|
|
|
FOR ADDITIONAL SAMPLES, USE ADDITIONAL COPIES OF PAGE 2. |
DA FORM 7539, FEB 2005 |
APD 9V1.000 |
|
PAGE 2 OF 2 |